Certifications Guide for Electric Toothbrush OEM: CE, FDA, FCC & More (2026)
An electric toothbrush OEM requires mandatory certifications for each target market: CE + RoHS for the EU (cost: $3,000–$8,000, 4–8 weeks), FCC for the US (cost: $1,500–$4,000, 2–4 weeks), PSE for Japan, KC for South Korea, EAC for Russia. IPX7 waterproof is the industry standard (not legally required). CE covers all 27 EU states; FCC covers the US only. The IEC CB Scheme allows one test report to certify for multiple markets, saving 30–50% on total costs when targeting 3+ regions.

Executive Summary: Electric Toothbrush OEM Certifications
- EU Mandatory (CE): Electric toothbrushes sold in the EU must comply with LVD (2014/35/EU), EMC (2014/30/EU), RoHS (2011/65/EU), and RED (2014/53/EU for Bluetooth models). Cost: $3,000–$8,000 · Timeline: 4–8 weeks. CE covers all 27 EU member states plus EFTA.
- US Mandatory (FCC + FDA): FCC SDoC is required for all electric toothbrushes. FDA Class I device registration is mandatory for the factory — consumer brands without therapeutic claims are 510(k) exempt. Cost: $1,500–$4,000 (FCC) · FDA facility registration: $11,423/year.
- Multi-Market Efficiency: The IEC CB Scheme allows one set of test reports to be used as the basis for CE, PSE (Japan), KC (South Korea), and RCM (Australia) — saving 30–50% on total certification costs when targeting 3+ markets.
- Key Insight: ODM sourcing can save $5,000–$15,000 in certification costs by leveraging the factory's pre-existing CE, FCC, RoHS, and IPX7 test reports on their existing platform.
目录
- Quick Reference: All Certifications at a Glance
- CE Marking (European Union)
- FCC Certification (United States)
- FDA Requirements (United States)
- RoHS & REACH (Environmental)
- Asia-Pacific: PSE, KC, EAC & Others
- IPX7 Waterproof Rating
- ISO Standards for OEM Factories
- Certification Cost Strategy for Multi-Market Brands
- 常见问题
1. Quick Reference: All Certifications at a Glance
Before diving into each certification in detail, here's the complete reference table for electric toothbrush OEM manufacturing:
| 认证 | 市场 | 类型 | 典型成本 | 时间表 | Required? |
|---|---|---|---|---|---|
| CE (LVD + EMC) | 欧洲联盟 | Safety + EMI | $3,000–$8,000 | 4-8 周 | Mandatory |
| CE RED | EU (Bluetooth models) | Radio | $2,000–$5,000 | 4-6 周 | If Bluetooth |
| FCC (SDoC) | 美国 | EMI | $1,500–$4,000 | 2-4 周 | Mandatory |
| FDA 注册 | 美国 | Medical device | $5,000–$15,000* | 4-12 周 | Required* |
| RoHS | EU / Global | Environmental | $500–$2,000 | 2-4 周 | Mandatory (EU) |
| REACH | 欧洲联盟 | Chemical safety | $1,000–$3,000 | 3-6 周 | Mandatory |
| PSE (Japan) | 日本 | 安全 | $2,000–$5,000 | 4-8 周 | Mandatory |
| KC (South Korea) | 韩国 | Safety + EMI | $2,000–$5,000 | 4-8 周 | Mandatory |
| EAC (Russia/CIS) | Russia, Belarus, Kazakhstan | 安全 | $2,000–$4,000 | 6–10 weeks | Mandatory |
| IPX7 | 所有市场 | Waterproof | $200–$800 | 1-2 周 | Industry standard |
| ISO 9001 | 所有市场 | Factory quality | $3,000–$8,000 | 6-12 个月 | Expected by buyers |
* FDA costs and requirements vary based on classification. Most consumer electric toothbrushes without therapeutic claims are Class I 510(k) exempt but still require facility registration.
CE + RoHS
Required to sell anywhere in the European Union — 27 member states plus EFTA.
FCC + FDA Reg
Required to sell in the United States. FCC for electromagnetic, FDA for device registration.
PSE + KC
Required for Japan and South Korea — high-value oral care markets with premium pricing.
ISO 13485
Medical device quality management. Differentiates your factory and supports clinical-positioned brands.

📋 About the Manufacturer: Relish Technology
Relish Technology is a Shenzhen-based oral care OEM/ODM factory with 14+ years of export experience in the electric toothbrush industry. Our existing platforms already hold CE, FCC, RoHS, IPX7, and PSE certifications — meaning ODM and private label buyers can often leverage existing test reports, saving months of certification time and $5,000–$15,000 in testing costs. Our facility: 20,000 m² workshop · 1.2M+ units/month · ISO 9001, ISO 13485 certified · Products exported to 120+ countries. Request a certification consultation →
2. CE Marking (European Union)
CE marking is the single most important certification for any electric toothbrush brand selling in Europe. Without it, your products cannot legally be sold in the EU, and customs authorities can seize and destroy non-compliant shipments at the border.
Applicable EU Directives
Electric toothbrushes fall under multiple EU directives simultaneously [1]. Your product must comply with all that apply:
- LVD (2014/35/EU) — Low Voltage Directive: Safety requirements for electrical equipment operating between 50–1000V AC. Tests electrical insulation, leakage current, mechanical strength, and temperature resistance. Standard: EN 60335-1 (general) + EN 60335-2-52 (specific to oral hygiene devices).
- EMC (2014/30/EU) — Electromagnetic Compatibility: Ensures the toothbrush doesn't emit excessive electromagnetic interference and is resilient to external EMI. Standards: EN 55014-1 (emissions) + EN 55014-2 (immunity).
- RoHS (2011/65/EU) — Restriction of Hazardous Substances: Limits the use of lead, mercury, cadmium, hexavalent chromium, PBBs, and PBDEs in electronic components and solder.
- RED (2014/53/EU) — Radio Equipment Directive: Only required if your toothbrush has Bluetooth or wireless charging. Tests radio frequency emissions and safety. Additional cost: $2,000–$5,000.

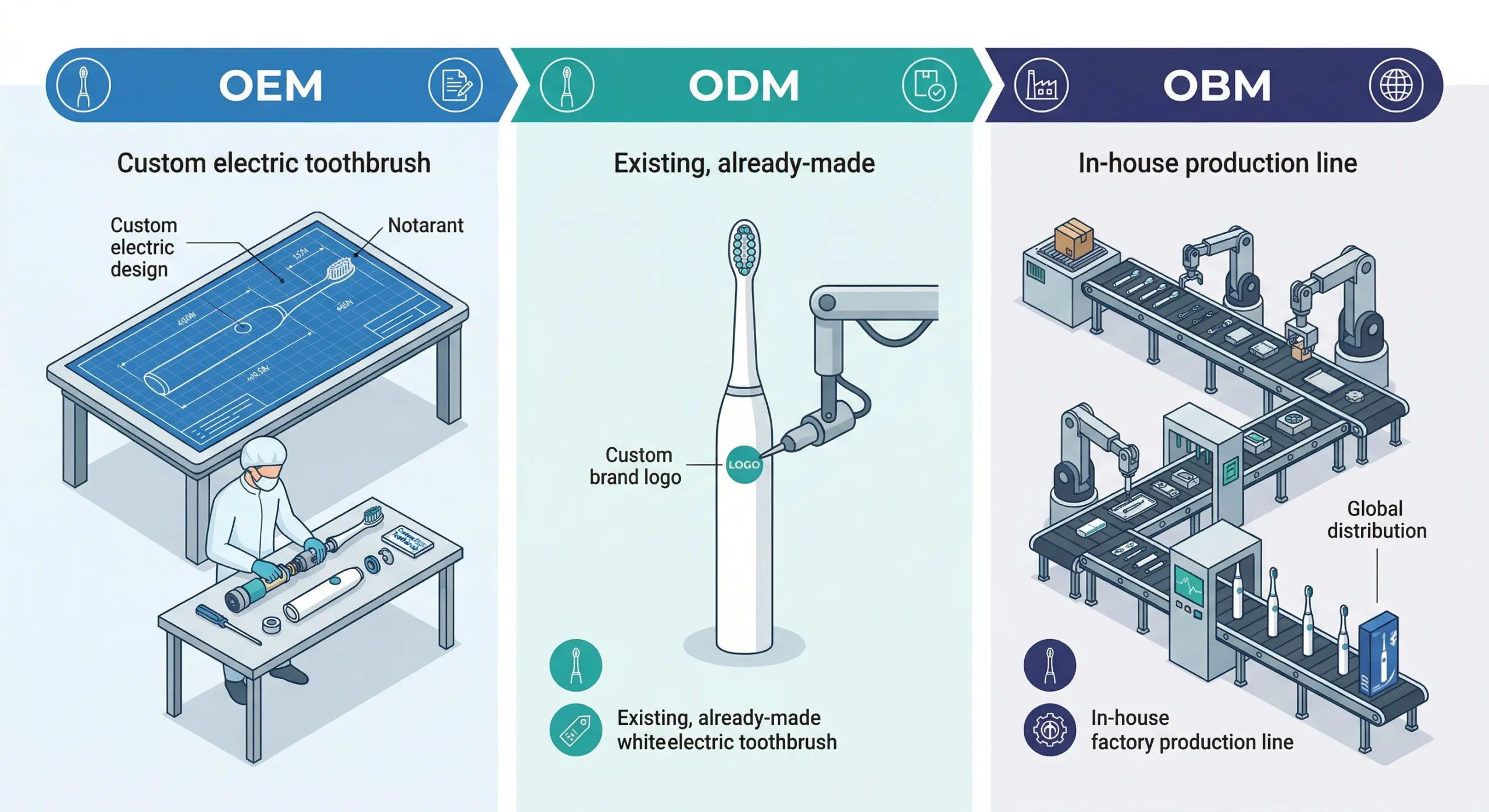
CE Certification Process
Identify applicable directives and standards
Determine which EU directives apply based on your product's features (Bluetooth? wireless charging? battery type?). Map each directive to its corresponding EN standard.
Choose a Notified Body (for LVD)
LVD assessment for electric toothbrushes typically requires a Notified Body — an accredited EU-designated testing organization. Options: TÜV Rheinland, TÜV SÜD, SGS, Intertek, DEKRA.
Submit samples for testing
Send 5–10 production-representative samples to the chosen lab. Testing covers: dielectric strength, insulation resistance, leakage current, mechanical strength, abnormal operation, water ingress (IPX7), and EMC.
Receive test report + EU Declaration of Conformity
Upon passing all tests, the lab issues a test report. You (as the brand/manufacturer) then draft and sign the EU Declaration of Conformity (DoC).
Affix CE marking and prepare Technical File
Print CE mark on product, packaging, and user manual. Compile a Technical File containing test reports, risk assessment, design documents, and the DoC. This file must be available for inspection for 10 years after last production.
⚠️ Who is the "manufacturer" for CE purposes?
In the EU regulatory framework, the "manufacturer" is the entity whose name and address appear on the product and packaging. If your brand name is on the product, you are the responsible manufacturer — not the Chinese factory. This means the CE liability sits with you. Ensure your contract with the factory clearly defines who handles CE compliance and maintains the Technical File.
3. FCC Certification (United States)
FCC (Federal Communications Commission) certification is mandatory for any electronic device sold in the United States that emits electromagnetic energy — which includes every rechargeable electric toothbrush with a motor and battery.
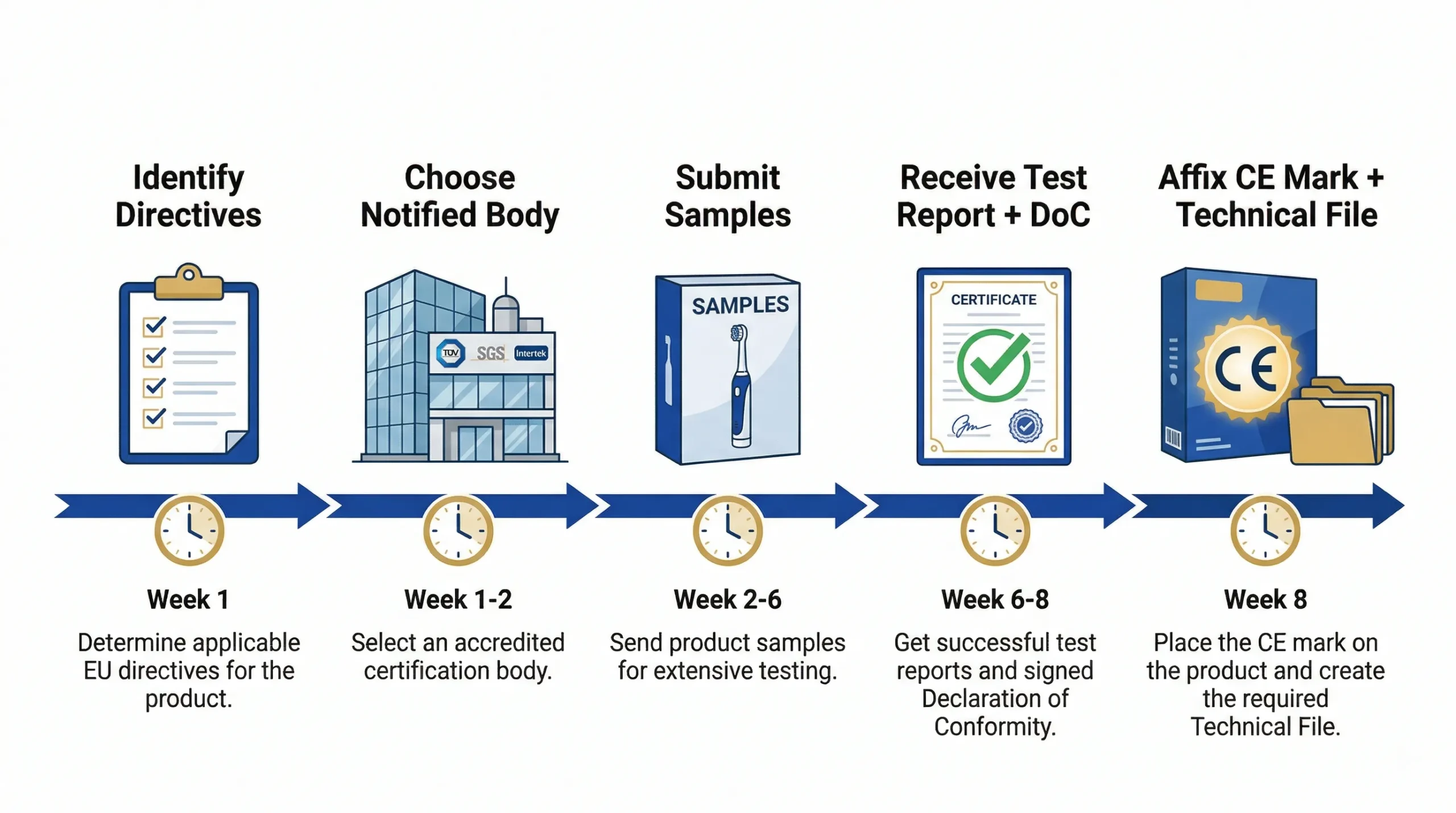
SDoC vs FCC ID
Most electric toothbrushes (without Bluetooth or wireless charging) qualify for SDoC (Supplier's Declaration of Conformity), which is the simpler path:
- SDoC: No FCC approval needed. You test at an accredited lab, receive a passing test report, and self-declare compliance. Affix the FCC logo + "This device complies with Part 15" statement on the label. Cost: $1,500–$4,000.
- FCC ID: Required if the device has intentional radiators (Bluetooth, Wi-Fi, wireless charging). Requires formal FCC approval through a Telecommunications Certification Body (TCB). Cost: $4,000–$10,000. Timeline: 4–8 weeks.
Testing Requirements
- Part 15.109: Radiated emissions — ensures the motor's electromagnetic emissions stay below FCC limits
- Part 15.107: Conducted emissions — tests emissions through the power supply / USB charging cable
- AC Power Line Conducted Emissions: If the device charges via AC adapter (not USB direct)
💡 ODM advantage: Pre-certified platforms
If you're sourcing via ODM, the factory's existing toothbrush platform may already have valid FCC SDoC test reports. You can leverage these reports for your branded version — as long as the motor, battery, and circuit board remain identical. This saves $1,500–$4,000 and 2–4 weeks. Confirm with your factory that the FCC report covers your exact configuration.
4. FDA Requirements (United States)
The FDA's role with electric toothbrushes is one of the most misunderstood areas in oral care manufacturing. Here's the reality:
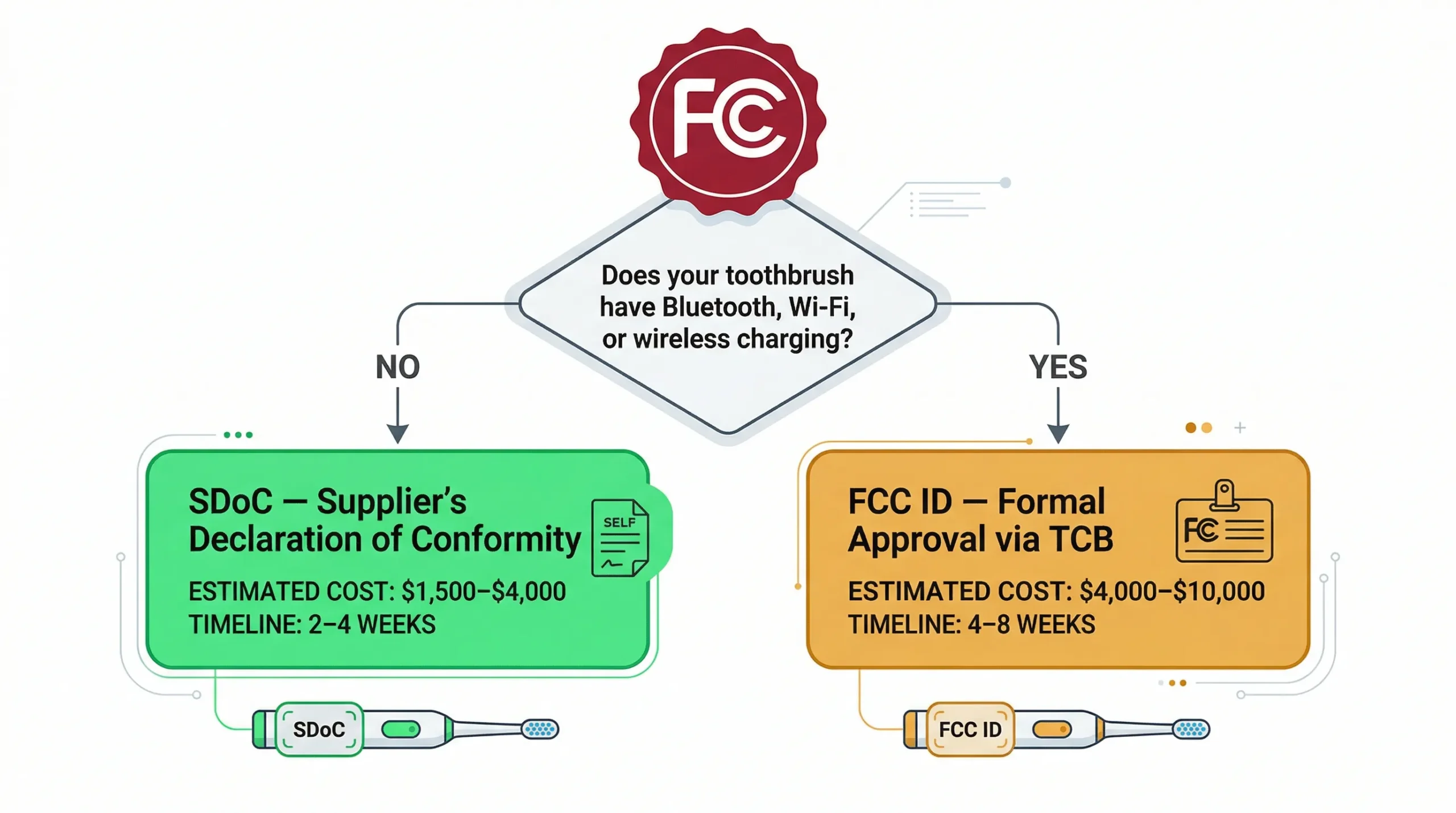
Classification: Class I Medical Device (510(k) Exempt)
The FDA classifies most consumer electric toothbrushes as Class I medical devices under 21 CFR Part 872 (Dental Devices), specifically 872.6840 (Powered toothbrush) [3]. They are generally exempt from 510(k) premarket notification IF no therapeutic claims are made on the packaging or marketing materials.
What IS required
- Facility Registration: Any manufacturing facility that produces medical devices for the US market must register with the FDA. The Chinese factory handles this — but the brand must also register as an "initial importer." Annual facility registration fee (FY2026): $11,423.
- Device Listing: Each product model must be listed in the FDA's device database, linked to the registered facility.
- Quality System (21 CFR Part 820): For Class I exempt devices, full QSR compliance is not mandatory, but the FDA expects "adequate quality controls." Most serious OEM factories have ISO 13485 or at minimum ISO 9001.
- Adverse Event Reporting (MDR): If your product causes injury, you must file a Medical Device Report with the FDA within 30 days.
When 510(k) IS required
- Your product makes therapeutic claims: "treats gingivitis," "clinically proven to reverse periodontal disease," "FDA cleared"
- Your product uses a significantly different technology than predicate devices on the market
- You want to market as a medical device for insurance reimbursement or professional dental use
If 510(k) is required, add $20,000–$100,000 和 3–12 months to your timeline.
⚠️ Don't say "FDA approved" for electric toothbrushes
The FDA does not "approve" Class I medical devices — it registers them. Using "FDA Approved" on marketing materials for an electric toothbrush is a compliance violation. Correct language: "FDA Registered" or "FDA Listed." This distinction matters for FTC advertising compliance.
5. RoHS & REACH (Environmental Compliance)
RoHS (Restriction of Hazardous Substances)
RoHS restricts the concentration of hazardous materials in electronic products sold in the EU [1]. The directive currently restricts ten substances in electronic equipment, including lead, mercury, cadmium, hexavalent chromium, PBB, PBDE, and four phthalates (DEHP, BBP, DBP, DIBP) [2]. For electric toothbrushes, the most critical limits are:
- Lead (Pb): < 0.1% (1000 ppm) — most critical for solder and PCB components
- Mercury (Hg): < 0.1% — relevant for battery cells
- Cadmium (Cd): < 0.01% (100 ppm) — relevant for battery contacts and plating
- Hexavalent Chromium (Cr6+): < 0.1% — metal finishes
- PBB / PBDE: < 0.1% — flame retardants in plastics
REACH (Registration, Evaluation, Authorization, Restriction of Chemicals)
REACH requires that any chemical substance manufactured or imported into the EU in quantities >1 ton/year must be registered with ECHA (European Chemicals Agency). For electric toothbrush brands, REACH primarily affects:
- Plastic housing materials (ABS, PP, TPE)
- Brush head bristles (nylon filaments)
- Adhesives and sealants used in assembly
- Battery electrolytes
Your material suppliers should provide REACH-compliant declarations. The OEM factory is responsible for compiling the compliance documentation, but the liability sits with the brand whose name is on the product.
💡 ODM shortcut: Pre-compliant materials
Established ODM platforms typically already use RoHS-compliant materials and have REACH declarations from their suppliers. Ask your factory for existing RoHS test reports and REACH SVHC (Substance of Very High Concern) screening reports before paying for new testing.

6. Asia-Pacific: PSE, KC, EAC & Others
Japan and South Korea are among the highest-value oral care markets globally — Japanese consumers pay $50–$150 for premium electric toothbrushes. But both markets have strict import certification requirements.
🇯🇵 PSE Mark (Japan)
Electric toothbrushes fall under Japan's Electrical Appliances and Materials Safety Act (DENAN). They require PSE (Product Safety Electrical Appliance & Material) certification.
- Category: Likely classified as "Electrical appliances for personal care" — requires菱PSE (diamond mark) which needs a registered certification body
- Standard: J60335-2-52 (Japan's adoption of IEC 60335-2-52)
- Process: Testing at a Japan-recognized lab + factory inspection (some CB scheme reports can be leveraged)
- Cost: $2,000–$5,000
- Timeline: 4-8 周
🇰🇷 KC Mark (South Korea)
South Korea requires KC (Korea Certification) for electric toothbrushes under the Korean Electrical Appliances Safety Control Act.
- Standard: K60335-2-52 (Korean adoption of IEC standard)
- Process: Testing at a KTL or KTC accredited lab. Some IEC CB reports accepted with additional national deviation testing.
- Cost: $2,000–$5,000
- Timeline: 4-8 周
🇷🇺 EAC Mark (Russia / EAEU)
The Eurasian Economic Union (Russia, Belarus, Kazakhstan, Armenia, Kyrgyzstan) requires EAC (Eurasian Conformity) marking for electrical appliances.
- Standard: GOST/TR CU 004/2011 (low-voltage equipment) + GOST/TR CU 020/2011 (EMC)
- Process: Samples tested at accredited lab in EAEU member state + factory audit for serial production certificates
- Cost: $2,000–$4,000
- Timeline: 6–10 weeks
🇦🇺 RCM Mark (Australia / New Zealand)
Australia and New Zealand share the RCM (Regulatory Compliance Mark) system.
- Standard: AS/NZS 60335.2.52
- Process: Testing + registration on the national database. IEC CB reports are generally well-accepted.
- Cost: $1,500–$3,000
- Timeline: 3-6 周

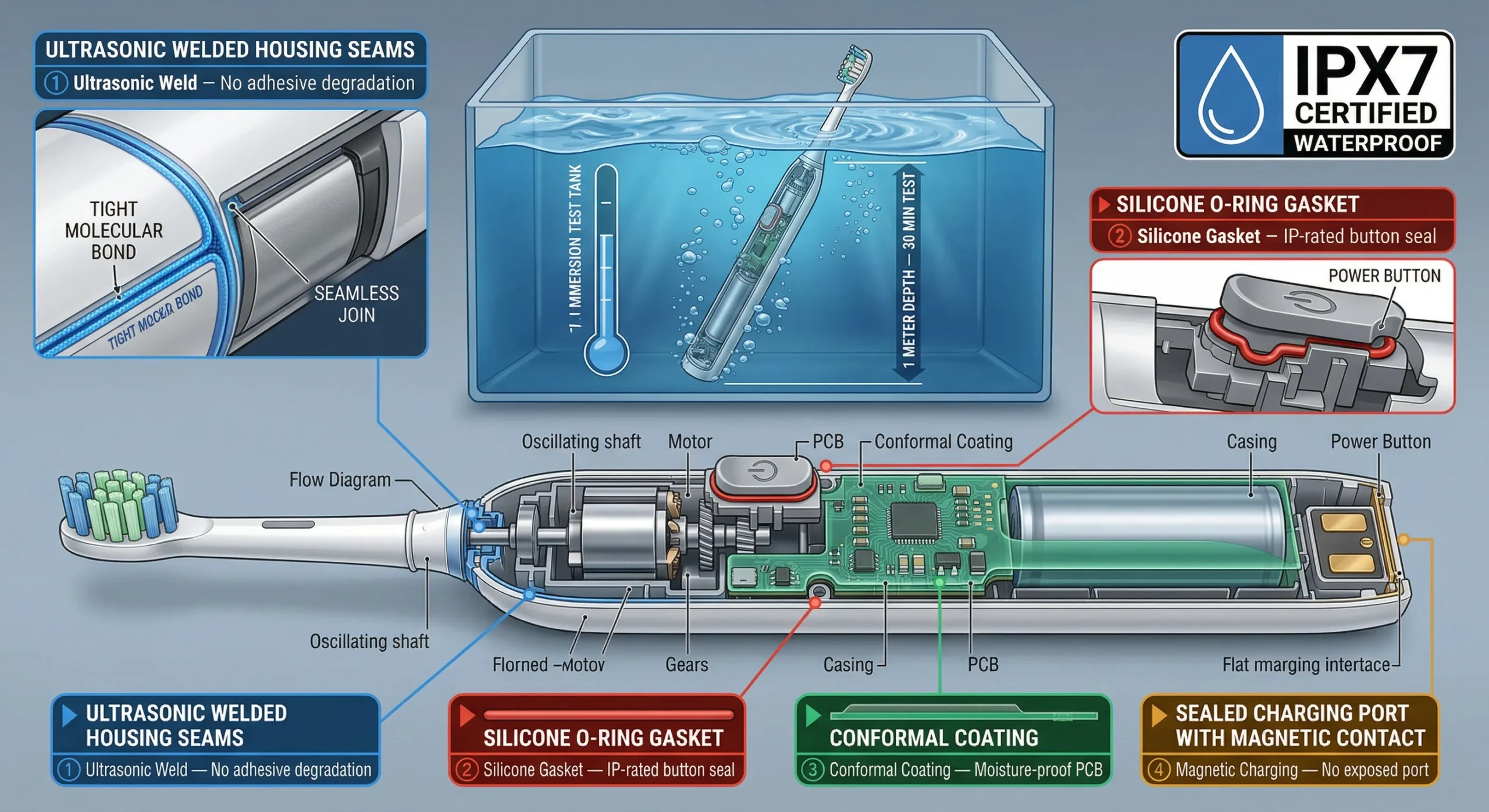
7. IPX7 Waterproof Rating
IPX7 is not a government-mandated certification — it's an international standard (IEC 60529) that has become the de facto minimum requirement for electric toothbrushes in all markets. Consumers expect to use their toothbrush in the shower and rinse it under running water.
What IPX7 means
- The device can withstand immersion in water up to 1 meter for 30 minutes
- Testing involves full submersion of the powered-on device in a water tank
- After immersion, the device must function normally with no water ingress detected
How OEM factories achieve IPX7
- Ultrasonic welding of the main housing seams (not adhesive — adhesive degrades)
- Silicone gaskets at all button interfaces and charging ports
- Conformal coating on the PCB to prevent corrosion from moisture
- Screw sealing with O-rings on the battery compartment (if serviceable)
Testing and cost
IPX7 testing costs $200–$800 per model at an accredited lab. Timeline: 1–2 weeks. Most reputable OEM factories have in-house IPX7 testing equipment and test 100% of production units at final QC stage (FQC). Ask your factory about their waterproof test coverage rate — 100% inline testing is the standard you should expect.
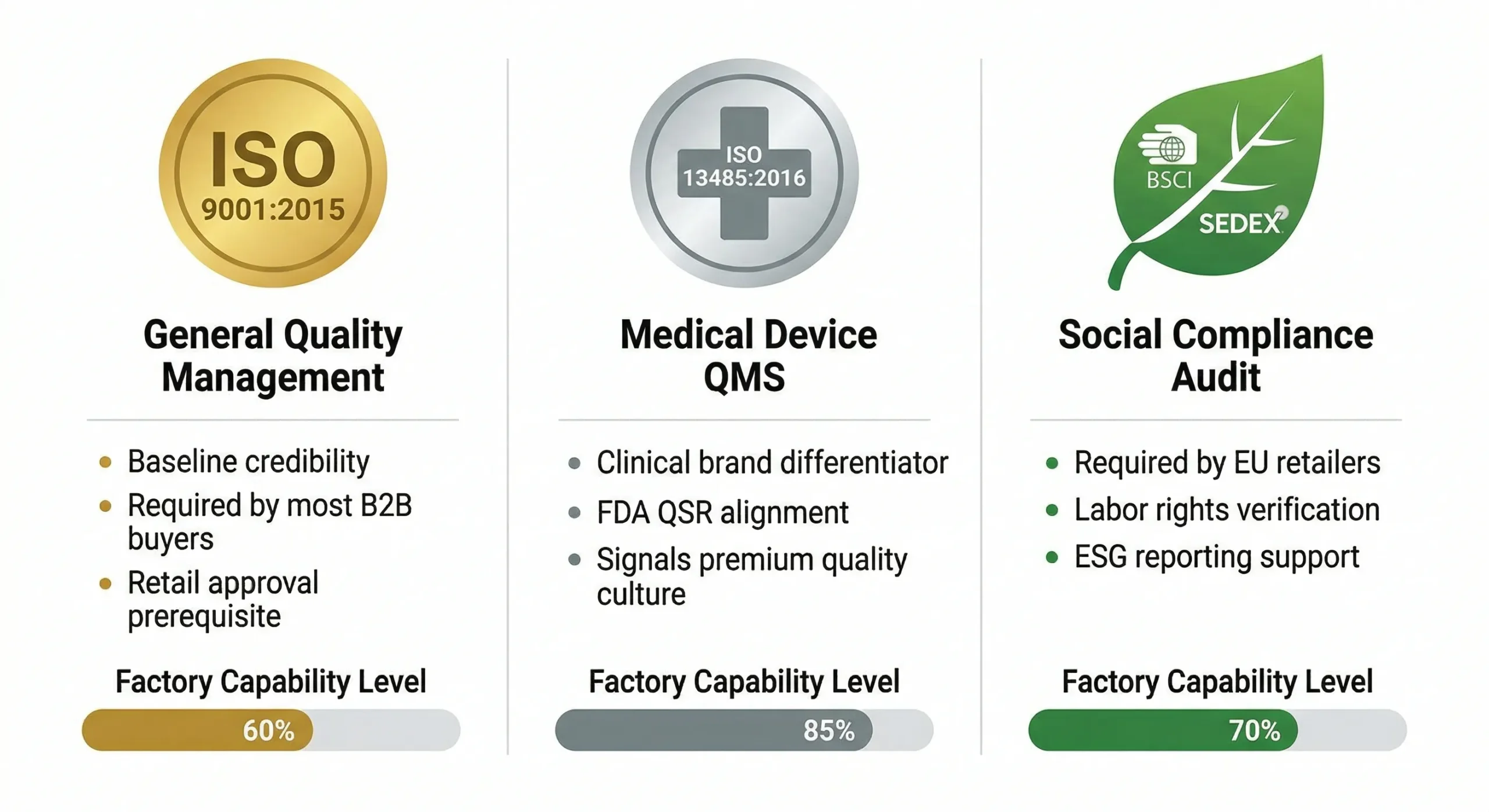
8. ISO Standards for OEM Factories
ISO certifications apply to the factory, not the product. But as a brand owner, your factory's ISO credentials directly affect your buyer confidence, retail approval, and regulatory risk profile.
| 标准 | What It Covers | Why It Matters for Your Brand |
|---|---|---|
| ISO 9001:2015 | General quality management system (QMS) | Expected by most B2B buyers. Required for retail buyer approval. Baseline credibility. |
| ISO 13485:2016 | Quality management for medical devices | Strong differentiator for clinical-positioned brands. Required for FDA QSR compliance. Signals serious quality culture. |
| ISO 22716:2007 | Good Manufacturing Practice for cosmetics | Relevant if you manufacture oral care cosmetics (toothpaste, mouthwash) alongside hardware. |
| BSCI / SEDEX | Social compliance and ethical audit | Required by many EU retailers. Covers labor rights, working conditions, environmental practices. |
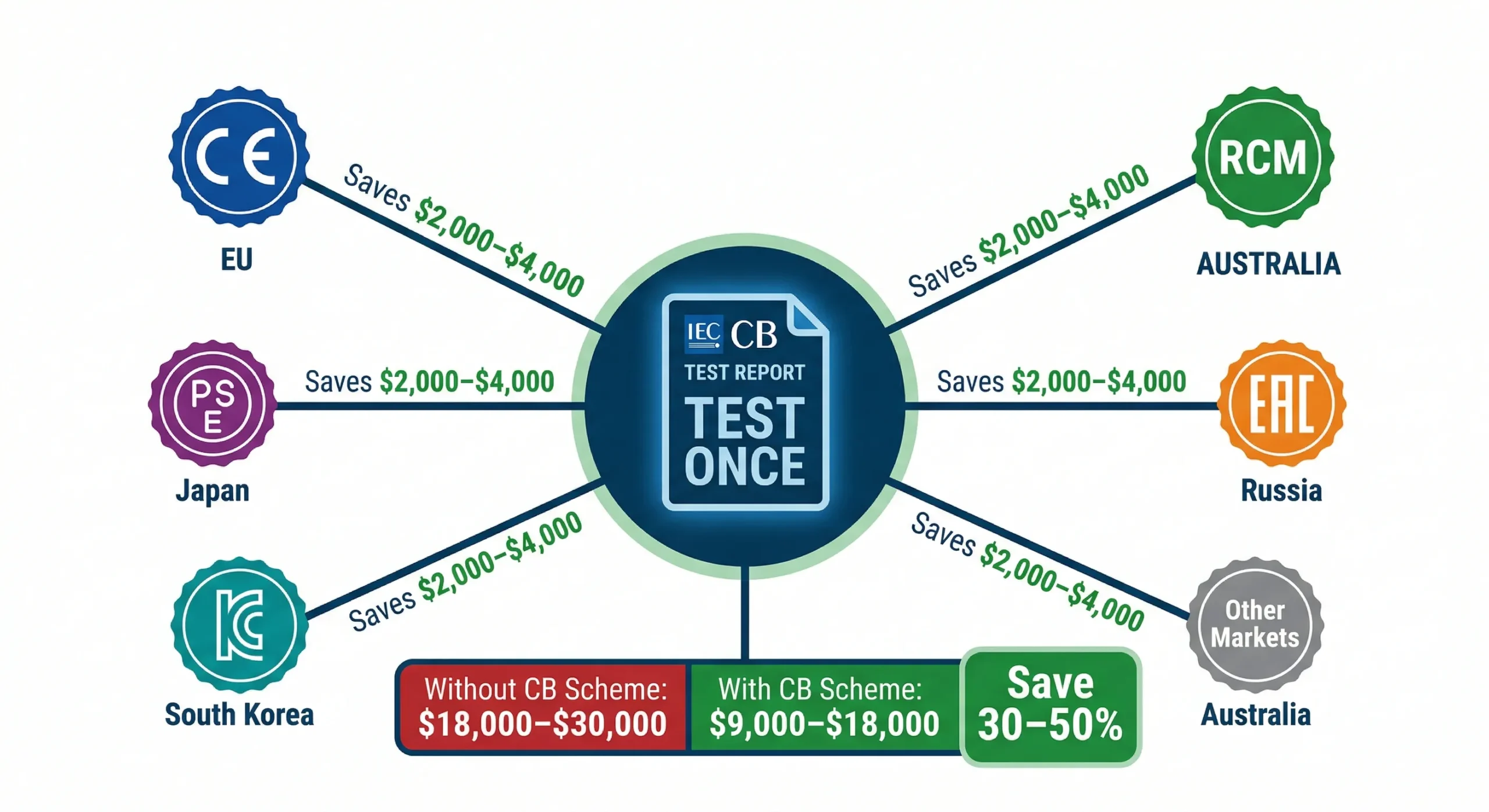
9. Certification Cost Strategy for Multi-Market Brands
If you're targeting more than one market, the total certification bill can quickly exceed $15,000–$25,000. Here's how experienced brands manage this efficiently:
The CB Scheme: Test Once, Certify Everywhere
"(《世界人权宣言》) IEC CB (Certification Body) Scheme allows you to run a single set of tests at one accredited lab, then use the CB test report as the basis for national certifications worldwide. This can save 30–50% on total testing costs when targeting 3+ markets.
Process: CB test report → CE (with Notified Body review) → PSE (Japan) → KC (Korea) → RCM (Australia). Each national body reviews the CB report and may run minor additional tests for local deviations.
Recommended certification sequence
Phased Certification Approach
- Phase 1 (Pre-production): CB test report + CE + FCC. Cost: $5,000–$12,000. This unlocks EU + US — your two largest markets.
- Phase 2 (First shipments): IPX7 test + RoHS test (if not bundled in CE). Cost: $500–$2,500.
- Phase 3 (Expansion): PSE (Japan) + KC (Korea) using CB report. Cost: $4,000–$10,000. Opens two premium markets.
- Phase 4 (As volume grows): EAC (Russia/CIS), RCM (Australia), BSCI audit. Cost: $5,000–$10,000.
💡 Key insight: ODM saves $5,000–$15,000 in certification costs
A quality ODM factory already holds CE, FCC, RoHS, and sometimes PSE/KC certifications for their existing platform. If your ODM product uses the exact same electronics and housing (just different branding), you can often leverage the factory's existing certifications — saving months of time and thousands of dollars. This is one of the strongest financial arguments for starting with ODM before investing in full OEM.
10.常见问题

Need Help with Certifications?
Relish Technology's existing platforms hold CE, FCC, RoHS, and IPX7 certifications. For ODM projects, you may leverage our existing compliance — saving months and thousands in testing costs. View client case studies →
Ask About Certification Support Read the Full OEM Guide参考资料
- [1] European Commission, Environment Directorate-General. (2024). RoHS Directive — Restriction of Hazardous Substances in Electrical and Electronic Equipment. .从 https://environment.ec.europa.eu/topics/waste-and-recycling/rohs-directive_en. Directive 2011/65/EU; restricts 10 hazardous substances including lead, mercury, cadmium, hexavalent chromium, PBB, PBDE, and four phthalates (DEHP, BBP, DBP, DIBP).
- [2] European Chemicals Agency (ECHA). (2024). Understanding REACH — Registration, Evaluation, Authorisation and Restriction of Chemicals. .从 https://www.echa.europa.eu/regulations/reach/understanding-reach. REACH entered into force on 1 June 2007, administered by ECHA in Helsinki.
- [3] U.S. Food and Drug Administration (FDA), Center for Devices and Radiological Health (CDRH). (2024). Medical Devices — Overview and Device Classification. .从 https://www.fda.gov/medical-devices. Electric toothbrushes classified under 21 CFR Part 872 (Dental Devices), 872.6840.